Advertisement
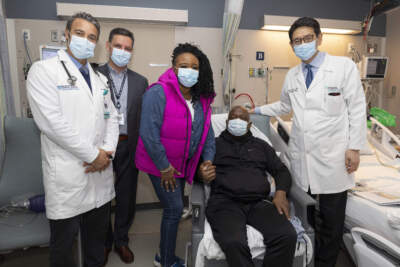
Richard Slayman, the first living patient ever to receive a genetically modified pig kidney as a transplant, walked out of Massachusetts General Hospital on Wednesday afternoon in good spirits, according...

Instead of automatically reporting to the state infants born dependent on addictive substances like opioids, new policies ask hospital staff to evaluate whether the newborn faces an imminent risk of...
Advertisement

The Boston Public Health Commission launched a campaign this week aimed at increasing awareness about colon cancer screening and the ways to combat one of the few known preventable cancers....

The hearing about private investments in health care focused primarily on the financial challenges facing Steward Health Care, one of the nation's largest private, for-profit hospital chains, and the uncertain...

Researchers at Massachusetts General Hospital and the University of Pennsylvania revved up immune cells that shrank an extremely aggressive type of brain tumor when tested in a handful of patients....

The report, from the Disability Law Center, found patients at Bridgewater State Hospital were subjected to illegally forced medication injections, violence from staff, lack of medical care, inappropriate medication for...